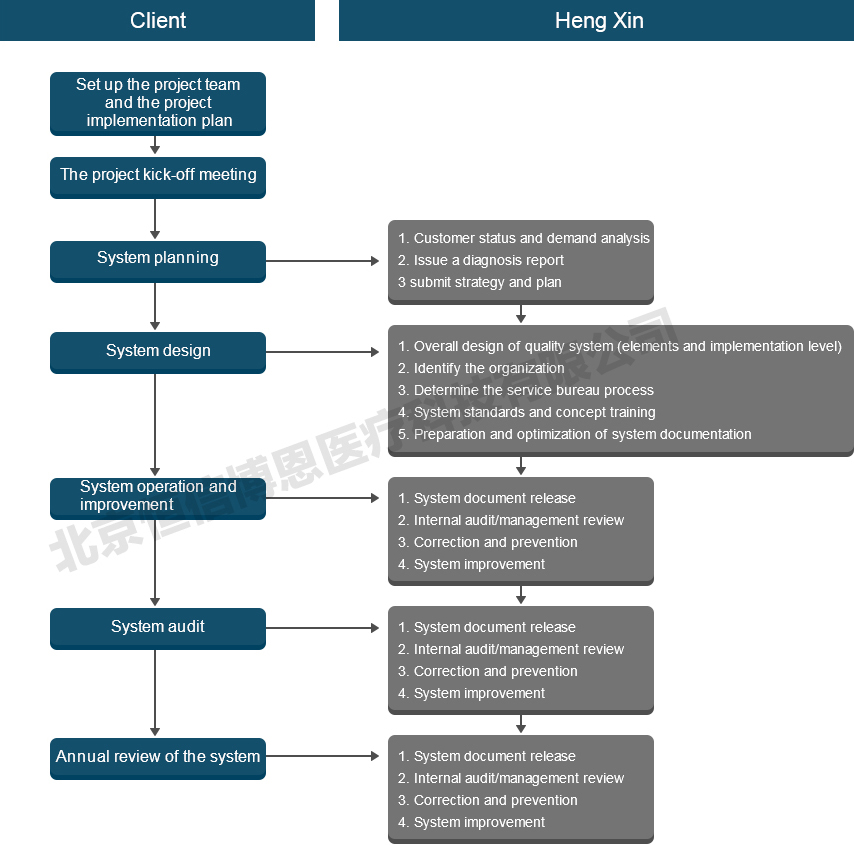
Project introduction
ISO 13485 is for medical equipment enterprise quality management systems. ISO 13485 is approved by countries all over the world through CE certification, United States FDA factory inspection, Japan J - the basis of the GMP and Canada CMDCAS, trust burns consulting in each big enterprise is widely recognized in domestic enterprises and certification company. The advisory services include:
Field research and diagnosis, evaluation of quality management status;
Standard training and internal training;
Guidance quality management system planning;
Guidance quality management system documentation;
To guide the quality management system operation and improve;
To guide the quality management system internal audit and management review;
Help choose certification institution and the third party certification related matters.
ISO13485 Authentication